Services
Parylene Coating

What is Parylene
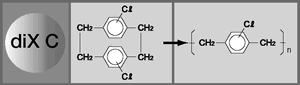
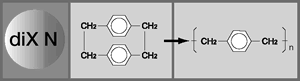
Parylene is the generic name for the poly-para-xylylenes. These materials form linear, highly-crystalline polymers but can be usefully produced only as coatings and films. The most commonly used is Parylene C, the mono-chloro substituted compound. Parylene N, the un-substituted compound, has better high-frequency dielectric properties, better penetrating power for coating the bore of very small diameter tubes, and is often preferred in medical applications. Parylene D, the di-chloro-substituted compound, has better high temperature endurance.
The Parylene Coating Process
The Parylene process is unique in coating technology and is best described as a vapor deposition polymerization. It is carried out under vacuum and requires specialized equipment.
- The process begins with sublimation at about 150°C of the high purity crystalline dimer di-p-xylylene.
- The vapor is pyrolised at about 650°C to form the gaseous monomer which has an olefinic structure.
- The coating chamber is at room temperature. The vapor condenses on all surfaces equally and can pass through holes as small as 1μ. It then spontaneously polymerizes to form a product with a high degree of crystallinity. The coating is absolutely conformal and can be laid down in thicknesses from a few angstroms to 50 microns or more depending on the requirements of the end use.
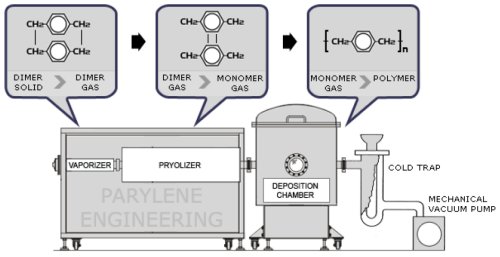
Parylene is applied at room temperature with specialized vacuum deposition equipment that permits control of coating rate and thickness. The deposition process takes place at the molecular level as the chemical, in dimer form, is converted under vacuum and heat to dimeric gas; pyrolized to cleave the dimer; and finally deposited as a clear polymer film.
The material is applied at .0002-in per hr. Coating thicknesses from .100 to 76 microns can be applied in a single operation. Typical coating thickness for circuit boards is .00 I-in.
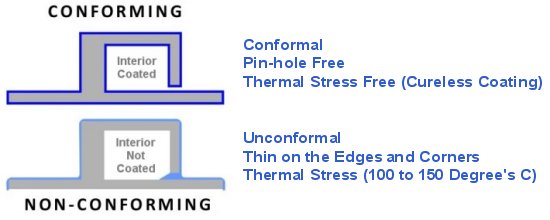
Because it is a gaseous process every surface is coated identically unless masked to prior specification. It is perfectly uniform over and inside of all surfaces. No other process can make this claim.
Parylene General Properties Benefits
- MIL-I-46058C, Type XY approved
- FDA approved – USP XXII, Class VI bio-compatibility rating
- UL listed
- Completely pin-hole free barrier coating
- Fully conformal on any type of surface material or design
- Inert transparent polymer
- Meets NBC requirements (ARlO / AFR80-38 / Navinst 3400.2)
- Barrier to oxygen, moisture, chemicals, solvents, and carbon dioxide
- Thermal mechanically stable between -200°C and 150°C
- Extremely high dielectric 5,000 volts per 0.001″ minimum
- Excellent adhesion properties
- Low stress coating that does not form sites prone to crack initiation
- Low / minimal impact on package cooling
- Hydrophobic
- Barrier to ionic and moisture species
- Chemical and fungal resistance
- Non-contaminating coating and coating process –no solvents, catalysts or other by-products are introduced during coating
- Entire process is accomplished
